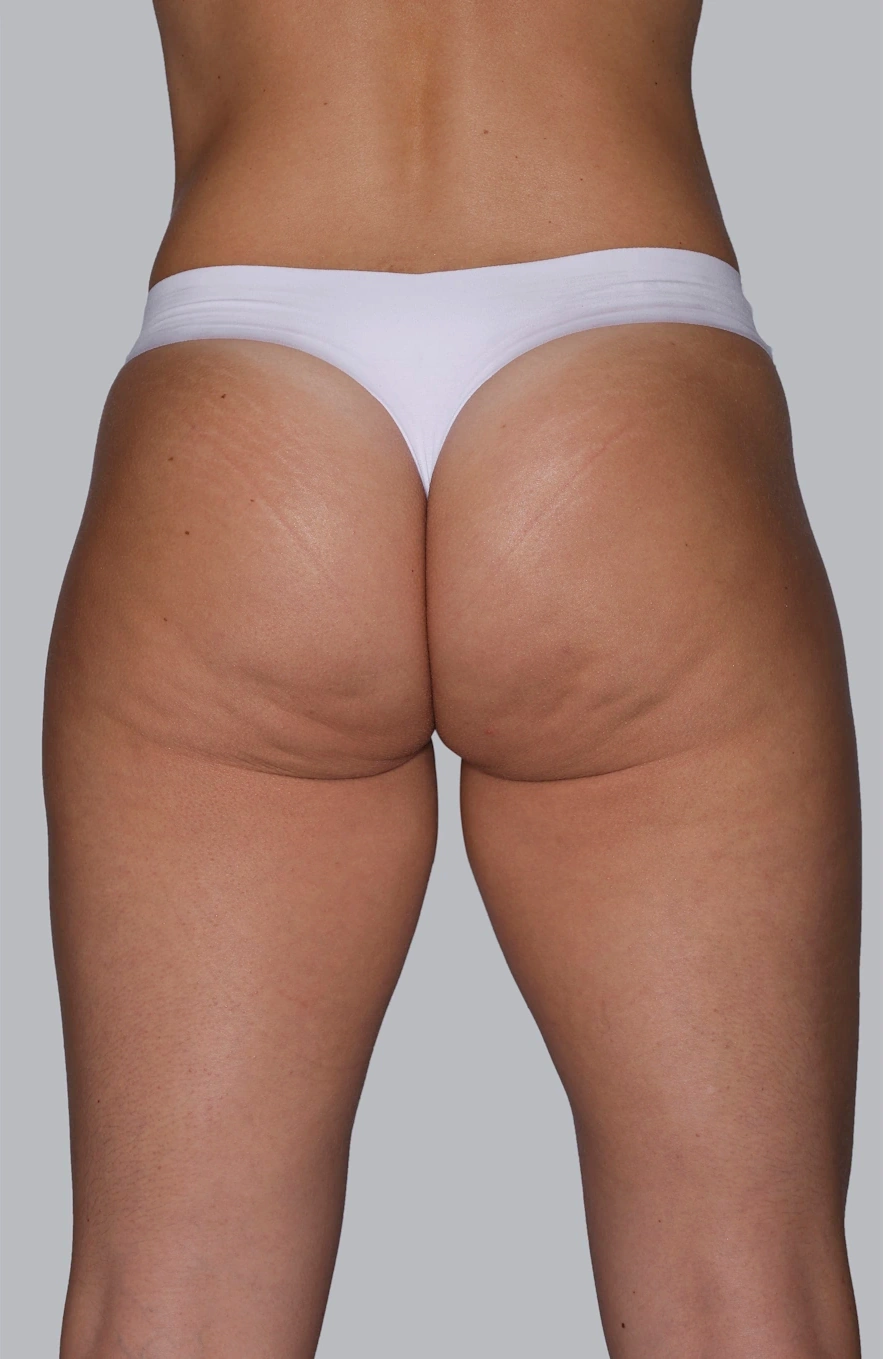
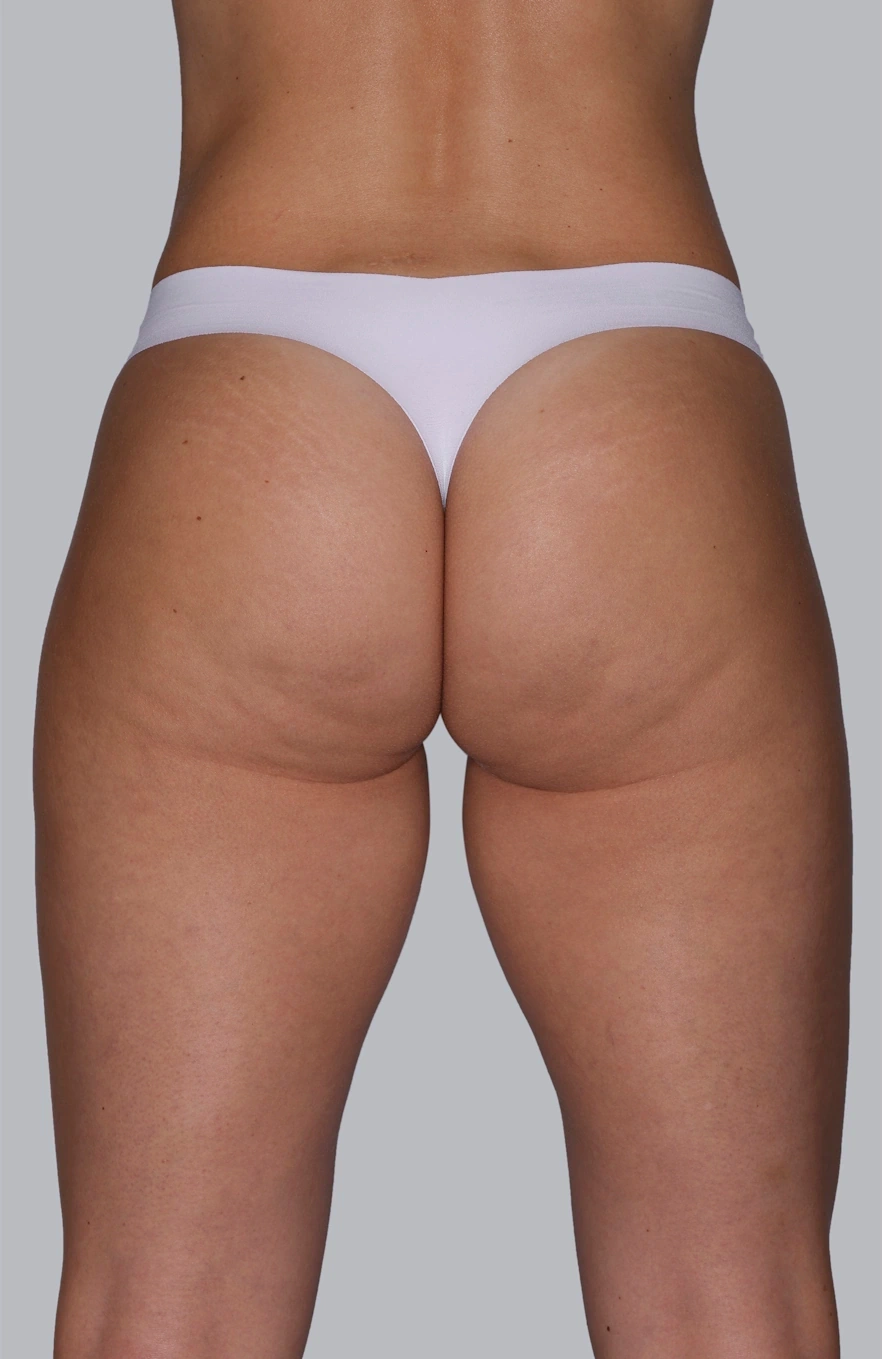
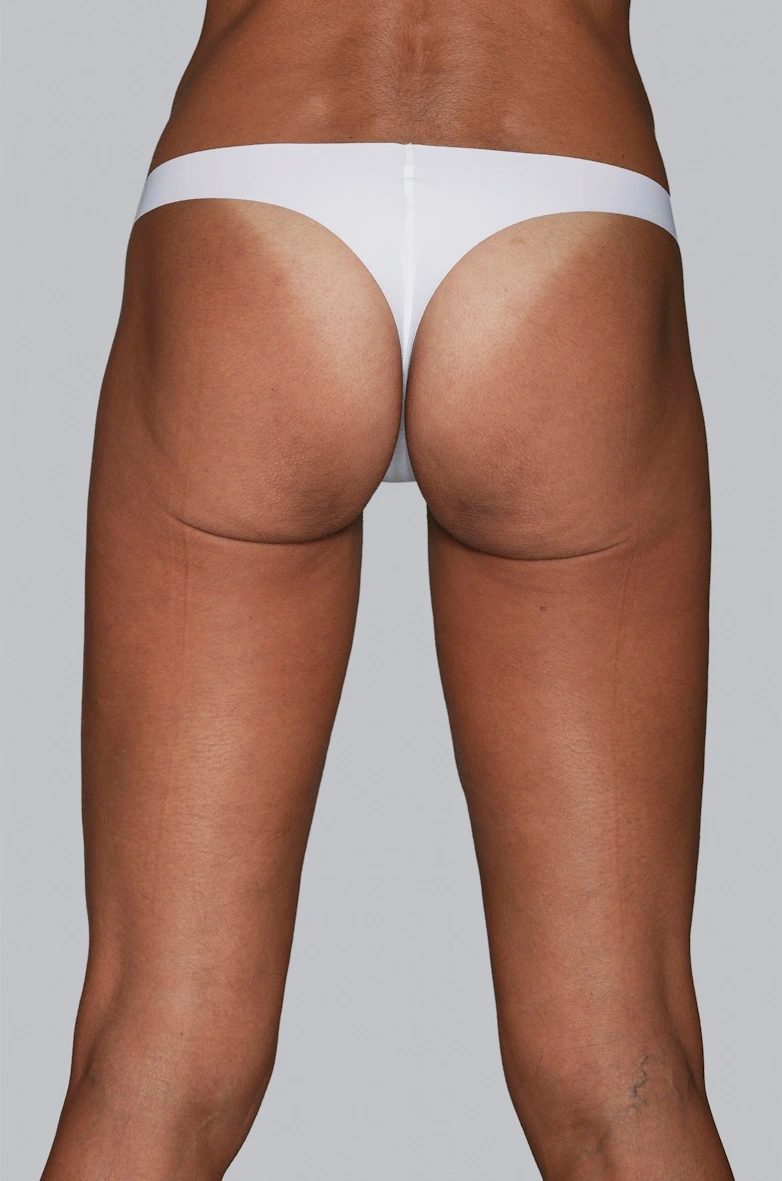
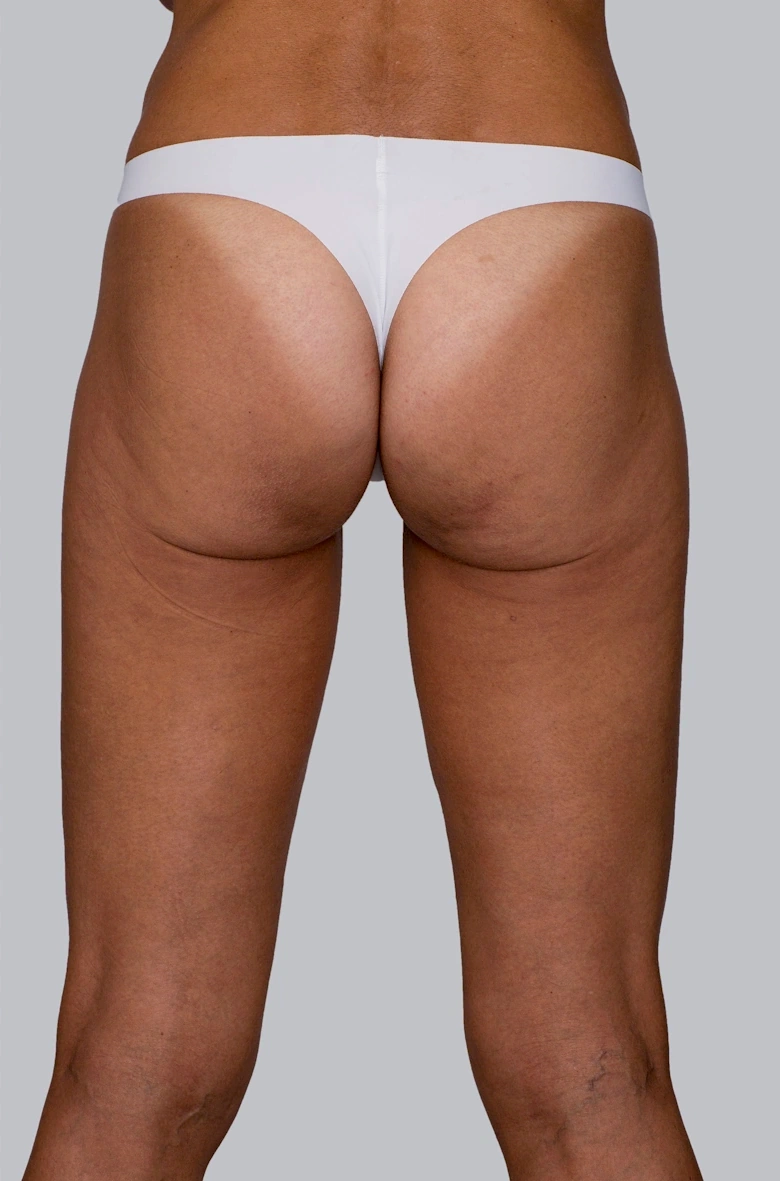
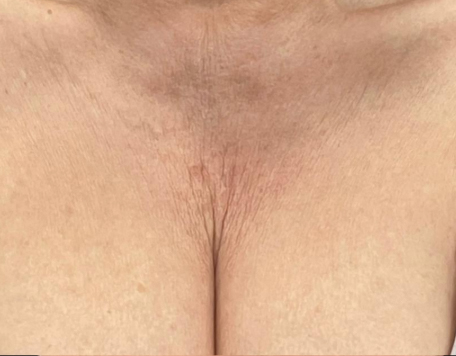
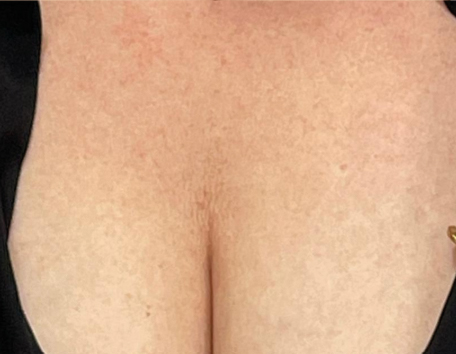
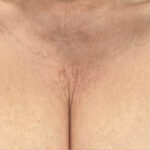
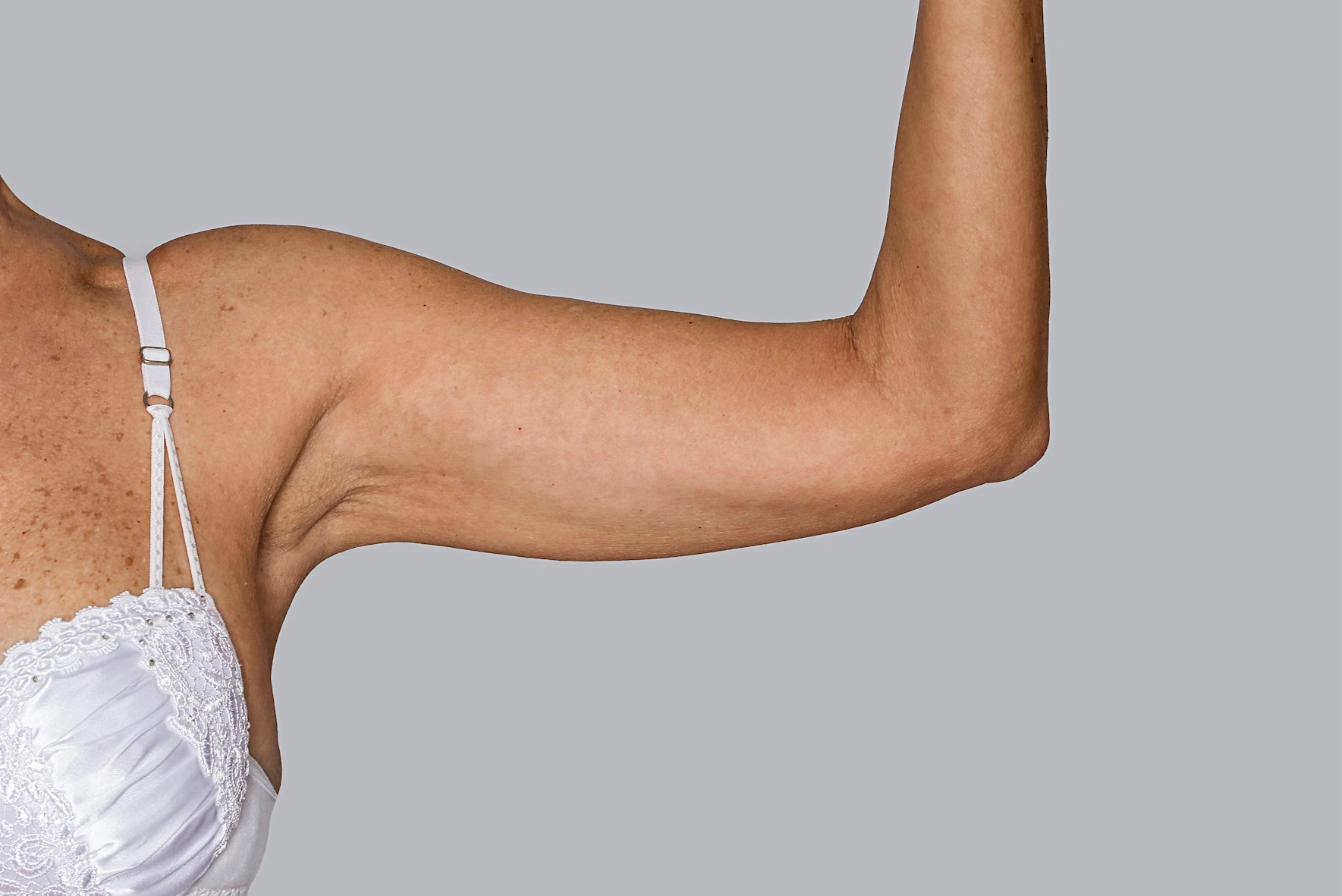Before & After photos
Real patient, individual results may vary

Shaping confidence that lasts
The first proven regenerative biostimulator
While Sculptra® has long been trusted for delivering natural-looking facial results, it has also been clinically proven to enhance areas of the body, including the buttocks, back of thighs, décolletage and upper arms. 5,18, 21,22,23
Sculptra® can be used across the body to improve firmness, smoothness, shape and contour. 1, 15-16, 18-19, 21, 23-25
By stimulating your body’s own collagen and elastin, it helps strengthen the skin’s foundation — with patients seeing improvements as early as one month after treatment, and continued improvement observed at 2 months and up to 1 year* (*cellulite of the posterior thighs)1, 9, 16, 18, 24, 26-27

“I always thought I could improve my buttocks, making them firmer and less saggy with a procedure and that’s how I chose Sculptra®.”
Renata, 36 years old

Real confidence. Real results.
‘‘I had my décolletage area done. It has just lifted the skin up. So, yeah I’m really happy with the results so far.’’
Gabriela, 36 years old

Frequently asked questions
Patients have been seeing improvements as early as one month after treatment, with continued improvement observed at 2 months and 1 year*18
*Cellulite in the posterior thighs
Sculptra® works progressively, so there will be gradual improvements in skin smoothness, elasticity, and contour over time.2,3,8,9,18,27
Is there any downtime after a Sculptra® treatment?
Good news—there’s minimal downtime with Sculptra®! You may experience mild bruising or bleeding, pain or tenderness, swelling, redness, or itching at the injection site, usually appearing soon after treatment. These effects are typically short-lived and resolve on their own within about a week, so most people return to their normal routine quickly.¹ Always consult your healthcare practitioner for personalized advice and to discuss safety information and potential risks associated with Sculptra®.
References
- Sculptra® MDR EU IFU
- Brandt FS, et al. Aesthet Surg J. 2011;31(5):521–528.
- Narins RS, et al. J Am Acad Dermatol. 2010;62(3):448–462.
- Widgerow A, et al. A randomized, comparative study describing the gene signatures of PLLA-SCA™ andCaHA in the treatment of nasolabial folds. Poster presented at IMCAS World Congress; 01–03 Feb 2024; Paris, France.
- Fabi S, et al. Effectiveness and Safety of Correction of Cheek Wrinkles Using a Biostimulatory Poly-L-Lactic Acid Injectable Implant. Poster presented at IMCAS World Congress; 26–28 Jan 2023; Paris, France.
- Huth S, et al. J Drugs Dermatol. 2024;23(4):285–288.
- Zubair R, et al. Dermatol Surg. 2024;50(12):1155-1162.
- Goldberg D, et al. Dermatol Surg. 2013;39(6):915–922.
- Bohnert K, et al. Plast Reconstr Surg. 2019;45(5):718–724.
- Triana L, et al. Aesthetic Plast Surg. 2024;48(20):4217–4227.
- El Hawa M, et al. J Cosmet Dermatol. 2025;24(1):e16624.
- Fares C, et al. J Cosmet Dermatol. 2025;24(2):e16671.
- Fabi S. Fillers for the Décolletage Area. https://assets.bmctoday.net/practicaldermatology/pdfs/PD0915_CF_Fillers.pdf [Accessed: October 2025].
- Gabriel A, et al. Aesthet Surg J Open Forum. 2023;5:ojad050. Published 2023 Jun 21.
- Sarubi J, et al. J Clin Aesthet Dermatol. 2023;16(6):30–36.
- Lin MJ, et al. Dermatol Surg. 2020;46(3):386–394.
- Mazzuco R, Sadick NS. Dermatol Surg. 2020;46:S86-S88
- Belezany k et al. Poster presented at IMCAS World Congress 01-03 February, 2024. Paris, France.
- Haddad A, et al. Plast Surg. 2025;49(5)1507-1517
- Data on File. (MA-55302). Sculptra Cellulite Pilot CSR 43CASA2006.
- Joseph J, et al. Poster presented at AMWC; 27–29 Mar 2024; Monte Carlo, Monaco.
- Nikolas A, et al. J Drugs Dermatol. 2022,21(3):295-303
- Mazzuco R, et al. J Cosmet Dermatol. 2022;21(12):6727-6733.
- Vanaman M, Fabi S. Plast Reconstr Surg. 2015;136(5 Suppl):276S-281S
- Wilkerson E, Goldberg D. J Cosmet Dermatol.2017;17(4):606-610.
- Galderma. Data on file (MA-57540).
- Mest DR, Humble G. Dermatol Surg. 2006;32(11):1336–134
Adverse events should be reported.
For the UK, Reporting forms and information can be found at www.mhra.gov.uk/yellowcard or search for Yellow Card in the Google Play or Apple App Store.
For Ireland, Suspected adverse events can be reported via HPRA Pharmacovigilance, Website: www.hpra.ie; Adverse events should also be reported to Galderma (UK) Ltd, Email: medinfo.uk@galderma.com, Tel: +44 (0) 300 3035674
UKI-SCU-2600006 DOP: April 2026
This material refers only to the product certified under Regulation (EU) 2017/745 (MDR). Legacy versions of the product remain available. All features and claims apply solely to the EU MDR-certified version. For full product information and the Instructions for Use (IFU) for the EU MDR-certified version, visit: www.galderma.com/library.